Gut health is one of the most overused terms in modern health writing, and one of the least precise. The problem is not that the gut has been overvalued. The problem is that it has been framed badly. If you want to improve gut health naturally, you need to understand what it actually means – not as a marketing term, but as a biological system with measurable criteria. The science is not thin. The translation is.
In medicine, gut health is not one magical function. Gastroenterologist Stephan Bischoff defined it as a state characterised by five measurable criteria: effective digestion and absorption, a stable microbiota, intact barrier function, effective immune activity, and the absence of significant gastrointestinal disease. The gut is not an isolated tube – it is a living ecosystem where microbes, epithelial cells, immune tissue, hormones, and nerve signalling are in constant communication. When that interface loses stability, the consequences rarely stay local.
For years, the gut was treated like plumbing. Now it is often treated like a master switch that explains everything. Both views miss the point. The gut matters not because it secretly controls the body, but because it is embedded in a wider biological network. It helps process food, regulate immune tolerance, maintain barrier integrity, and relay signals beyond digestion. That is a more useful way to understand gut health – and a more honest one.
Why Diet Matters More Than Gut Trends
Diet matters because it is one of the most repeated inputs this system receives. What you eat shapes the composition and function of the gut microbiome, changes the metabolites microbes produce, and influences the intestinal barrier and mucosal immune system. Food is not just fuel. It is substrate, signal, and instruction.
Fibre-rich, minimally processed diets tend to support microbial diversity and beneficial metabolites such as short-chain fatty acids. Diets built around low fibre and heavily processed foods tend to do the opposite, pushing the gut towards reduced diversity, barrier disruption, and chronic inflammatory pressure.
This is why the idea that one supplement or one “gut-healing” food can fix everything is so weak. Gut health is shaped less by isolated products than by repeated dietary pattern. The gut responds to patterns of pressure applied over time, not to nutritional theatre. The real question is not which single food heals the gut. It is what kind of diet keeps the gut microbiome, the barrier, and the immune system working together over time. That is where the real work to improve gut health naturally begins – not with a product, but with a pattern.
Dietary Fibre: Still The Centre Of Gravity
If there is one part of the conversation about how to improve gut health naturally that remains stubbornly clear, it is dietary fibre. That is partly because fibre is unglamorous. It does not lend itself to grand promises, come with the tidy logic of a single supplement or the marketing appeal of a performance nutrient; it simply does its job. And in gut biology, that job is extremely important.

Fibre is the part of plant food human enzymes do not fully break down in the small intestine. It reaches the colon largely intact, where it becomes substrate for the microbial side of the system. That distinction matters. Protein, fat, vitamins, and minerals feed the host. Fibre also feeds the ecology the host depends on.

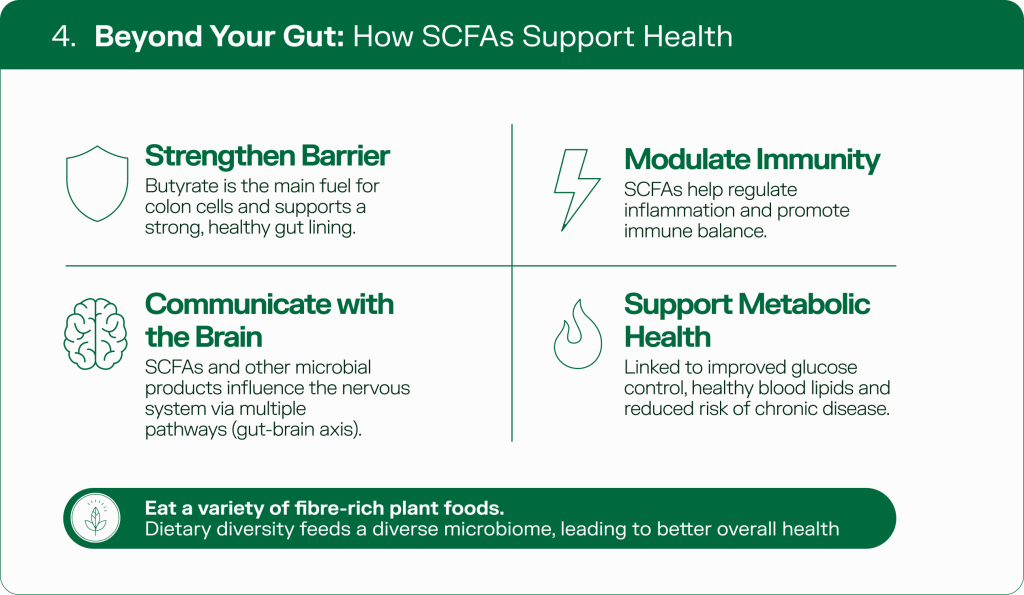
When that microbial fermentation is working well, it produces short-chain fatty acids such as acetate, propionate, and butyrate – estimated to occur in a molar ratio of approximately 60:20:20. These are not incidental by – products. They are part of how diet gets translated into gut function. Butyrate, in particular, is a primary fuel source for colon cells and helps maintain barrier integrity and immune balance. More broadly, short-chain fatty acids help regulate metabolism, inflammation, immune function, and cell proliferation across the gut environment.

This is also why low-fibre diets do more damage than people usually realise. When fibre intake stays low, microbial diversity tends to fall, short-chain fatty acid production drops, and the ecology begins to shift towards less favourable forms of metabolism. In experimental work using gnotobiotic mouse models, long-term deprivation of microbiota-accessible carbohydrates reduced diversity so severely that some microbial losses could not be recovered by simply adding fibre back later. Other work shows that fibre deprivation promotes the expansion of mucin-degrading bacteria such as Akkermansia muciniphila and Bacteroides caccae, thinning the mucus layer and making the barrier more vulnerable to pathogen invasion.
Modern diets make this worse. Across industrialised settings, fibre intake remains below recommended levels, while dietary patterns built around processed foods, saturated fat, refined carbohydrate, and low plant diversity continue to shape a less resilient microbiome. This helps explain why the search for foods that support gut health keeps circling back to the same foundations: legumes, vegetables, fruit, nuts, seeds, and whole grains. Not because they are trendy, but because they supply the substrates the system actually runs on.
And it is worth noting: different fibres favour different taxa. Different food matrices arrive with different combinations of starches, polyphenols, fats, micronutrients, and physical structures. That is why the best gut-friendly foods are usually ordinary foods eaten repeatedly and in variation, not a single overpraised ingredient.
Fibre is not the whole story of gut health. But it is still the nutritional centre of gravity.
The Practical Framework to Improve Gut Health Naturally
When it comes to how to improve gut health naturally in practice, most people obsess over what to eat. A highly effective, clinically sound starting structure is the 50-25-25 framework: half your plate dedicated to fibre-rich foods, a quarter to protein, and a quarter to carbohydrates.
A useful protein intake supports muscle mass, satiety, recovery, and metabolic health. The problem arises only when protein displaces fibre. A high-protein, low-carbohydrate diet significantly reduces total short-chain fatty acid and butyrate production, while increasing metabolites derived from amino acid fermentation. A high-fibre diet, by contrast, helps keep saccharolytic fermentation dominant and can counter many of the detrimental effects associated with heavy meat and fat intake.
Fermented foods – yoghurt, kefir, kimchi, chaas, idli, are also worth including regularly. They can introduce beneficial species directly and support microbial diversity. But they sit best within a broader structure that already has enough fibre and plant diversity. Treating them as a standalone solution is not supported by the evidence.
But the ratio is only half the story. The order in which you consume those foods can alter your glycaemic and metabolic response, even when the macronutrient quantities remain exactly the same. Sequence dictates signalling.

Fibre first. Consuming fibre first slows gastric emptying. It acts as a biochemical net, altering how quickly glucose enters your system. By preventing rapid glucose spikes upfront, you create a stable environment while simultaneously feeding the gut microbiome.
Protein second. Eating protein next enhances your body’s insulin signal. It improves how efficiently glucose is handled, stimulates incretin hormones like GLP-1, and triggers satiety hormones like PYY and CCK – all while providing the building blocks to preserve lean muscle mass.
Carbohydrates last. Carbohydrates are not the enemy. They provide immediate, usable energy, replenish liver and muscle glycogen, support thyroid function and metabolic rate, and improve serotonin production. But when consumed after fibre and protein, their metabolic impact is transformed. Glucose entry is significantly slower. The insulin response is efficient and controlled. Energy is sustained, not spiked.
When you combine fibre and protein before carbohydrates, you regulate insulin release far more efficiently. The clinical outcomes of this sequencing are meaningful: lower glucose variability, reduced metabolic stress, higher satiety signals, and better overall metabolic stability.
Same foods. Different sequence. Markedly better outcomes.







This sequencing also supports metabolic flexibility – your body’s ability to switch efficiently between burning glucose and burning fat for fuel. When the gut microbiome is well-fed and insulin signalling is stable, the metabolic switch stays responsive rather than getting stuck on one fuel source. Metabolic inflexibility – the inability to toggle between fuels – is often the first sign that something upstream has shifted, and it frequently begins with how and what you eat. Periodic time-restricted eating – such as a 12-to-14 hour overnight fast – can further support metabolic flexibility by training the body to access stored fat between meals, though this sits best within a dietary structure that already has adequate fibre and nutrient density.
This matters because most people do not need another rigid, extreme dietary religion. They need a sustainable framework – something they can repeat on a Tuesday, with real food, within their existing routine.
Note: This is general information for healthy individuals, not medical advice. If you have a medical condition or take medication, consult your clinician.
Gut Health and Longevity
Gut health is rarely discussed in the context of biological ageing, and that is a significant oversight. The mechanisms through which diet shapes the gut microbiome – SCFA production, barrier integrity, immune calibration, inflammatory tone – are not peripheral to the ageing process. They are embedded in it.
Chronic low grade inflammation, often called inflammageing, is one of the most consistent features of biological decline. The gut barrier is one of its primary sources: when barrier function weakens, bacterial endotoxins enter the bloodstream and trigger systemic inflammatory cascades that accelerate tissue damage across organs. The gut microbiome also intersects with metabolic regulation, insulin sensitivity, and cellular stress responses – all of which connect to the hallmarks of ageing.
Left uncorrected, this trajectory connects directly to metabolic syndrome – a cluster of at least three of five dysfunctions including elevated fasting glucose, excess visceral fat, high triglycerides, low HDL cholesterol, and high blood pressure. The gut microbiome intersects with every one of these markers through its influence on SCFA production, insulin sensitivity, and systemic inflammatory tone. Metabolic syndrome is a state of dysregulation, and dynamic systems can be corrected. But the correction starts upstream, with how you feed the gut.
The gut sits at a critical junction of the systems that determine your longevity. Feeding the microbiome is not separate from maintaining metabolic health, immune competence, and inflammatory resilience. It is the same project, viewed through a different lens. Emerging research in nutrigenomics – the study of how nutrients interact with gene expression – is beginning to map these connections more precisely, showing how dietary inputs influence not just microbial composition but the expression of genes involved in barrier integrity, inflammatory signalling, and metabolic regulation. To improve gut health naturally is, in that sense, an investment in how well the whole system ages.
Frequently Asked Questions: How to Improve Gut Health Naturally.
The collective community of approximately 38 trillion microorganisms – bacteria, fungi, viruses, and archaea – inhabiting the human gastrointestinal tract. The revised figure of 38 trillion replaced the older estimate of 100 trillion. These organisms perform metabolic functions the human genome cannot, including fibre fermentation, immune education, vitamin synthesis, and neuroactive compound production.
Gut health is a state characterised by five measurable criteria: effective digestion and nutrient absorption, absence of gastrointestinal illness, a normal and stable intestinal microbiota, effective immune status, and a general state of well-being.
Increase the diversity of dietary fibre (aim for 30 grams or more per day from multiple plant sources), eat fermented foods with live cultures regularly, reduce ultra-processed food intake, and include prebiotic-rich foods such as garlic, onions, leeks, and asparagus. These strategies work synergistically – they are not independent interventions.
Within 24 hours. However, rapid shifts are also reversible – the microbiome often rebounds when the original diet returns. Sustained changes in microbial composition and function typically require consistent dietary shifts over weeks to months.
SCFAs – acetate, propionate, and butyrate, are produced when gut bacteria ferment dietary fibre. Acetate is the most abundant (approximately 60 per cent of total SCFAs). Butyrate is the preferred fuel for colonocytes and has anti-inflammatory and barrier-strengthening properties. A fibre-deprived diet reduces SCFA production and can force bacteria to degrade the gut’s protective mucus lining – a direct pathway to increased intestinal permeability.
The evidence is more nuanced than the marketing suggests. Most commercial probiotic strains do not permanently colonise the gut. Their efficacy is strain-specific and depends on existing microbial composition. For most healthy people, regularly consuming fermented foods is a more ecologically coherent approach than supplementation. Probiotic supplements may have value in specific clinical contexts, such as post-antibiotic recovery.
Yes. The mechanisms through which diet shapes the gut microbiome – SCFA production, barrier integrity, immune calibration, inflammatory tone, are directly relevant to biological ageing. Chronic low-grade inflammation originating from a weakened gut barrier is a recognised driver of age-related tissue damage. The gut also intersects with metabolic regulation, insulin sensitivity, and cellular stress responses.
Increasingly real. Individual responses to the same foods vary based on existing microbial composition and genetic variation — the field of nutrigenomics studies exactly how nutrients interact with gene expression, including genes that influence gut barrier function, inflammatory response, and microbial metabolism. The universal principles – diverse fibre, fermented foods, reduced processed food – remain the foundation, but personalisation based on microbiome, metabolic, and nutrigenomic data is an emerging optimisation layer.